Newly FDA-approved microsurgical system holds promise for training, new treatments

Editor's Note The US Food & Drug Administration (FDA) has granted De Novo authorization for marketing MMI North America Inc.’s Symani Surgical System, according to the agency’s April 9 roundup. The system employs a motion scaling function to scale down surgeons movements for procedures requiring small instruments and/or microscopes, such…
FDA approves new antibiotic for staph, other infections

Editor's Note The US Food and Drug administration approved Zevtera for treating Staphylococcus aureus bacteremia (SAB) bloodstream infections, which are often acquired in healthcare settings. In the April 3 announcement, the agency notes the new antibiotic Zevtera (ceftobiprole medocaril sodium for injection) is effective for SAB-infected patients with right-sided infective…
FDA warning: unapproved topical analgesics potentially dangerous

Editor's Note The US Food and Drug Administration (FDA) advises consumers to avoid topical analgesics marketed in violation of federal law by six different companies, including: TKTX Company: TKTX Numb Maximum Strength Pain Reliever, Mithra+ 10% Lidocaine, TKTX During Procedure Numbing Gel 40% and J-CAIN cream [LIDOCAINE] 29.9% SeeNext Venture,…
Swifty’s singing keeps brain surgery on track

Editor's Note Staying awake during brain surgery to sing Taylor Swift songs helped ensure the best possible outcome for Selena Campione, a 36-year-old teacher from Stanhope, New Jersey who recently had a tumor removed at Jersey Shore University Medical Center. As detailed in a March 21 report from People, neurooncologist…
Procedural sedation analgesia considerations for ASC leaders
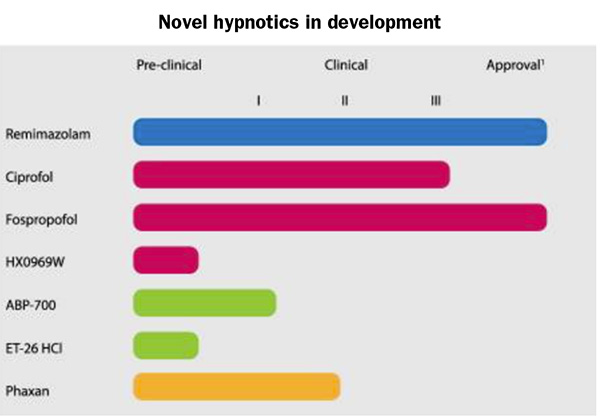
The promise of quicker recovery and fewer complications from sedation, anesthesia, and pain management have drawn clinicians and patients alike to procedures performed in ambulatory surgery centers (ASCs) and other outpatient settings. However, sedation, anesthesia, and analgesia add their own risks to those of the procedure itself. Understanding the latest…
FDA updates warning about plastic syringes made in China

Editor's Note An ongoing US Food and Drug Administration (FDA) evaluation of plastic syringes made in China reveals that quality problems are more widespread than initially believed, the agency reported on March 19. The update to an original November 23 safety notice recommends the following action to alleviate potential quality…
New EPA standards to reduce ethylene oxide emissions

Editor's Note New standards from The Environmental Protection Agency promise to cut nationwide emissions of ethylene oxide—employed to sterilize more than half of US medical devices—by more than 90 percent. According to a March 15 MedPage Today report, the aim is to reduce cancer risk among the 13 to 14…
FDA issues warning on fraudulent, unreliable medical device testing data
Editor's Note The FDA has issued a warning regarding fraudulent, duplicated and otherwise unreliable data coming from third party test labs that produce data for medical device companies, including many based in India and China. Issued February 20, the alert warns medical device manufacturers that they are responsible for confirming the…
FDA recalls BrightView SPECT systems
Editor's Note The FDA has issued a Class I recall of Philips’ BrightView, BrightView X and BrightView XCT Single Photon Emission Computed Tomography (SPECT) scanners due to a component failure that could result in the system’s detector falling on patients. Philips sent all affected customers an Urgent Medical Device Correction…
FDA announces urgent chest drain recall
Editor's Note The FDA has announced a recall of the Atrium Express Dry Suction Dry Seal Chest Drain, a disposable device used to remove air and/or fluid from the chest cavity or mediastinum and to aid in lung expansion and breathing. The drain is being recalled by maker Maquet Cardiovascular,…

 Free Daily News
Free Daily News