Editor's Note The Food and Drug Administration (FDA) on July 6 identified the recall by Getinge USA Sales Inc of its Flow-c and Flow-e Anesthesia Systems as Class I, the most serious. The recall was initiated after reports of cracked or broken on/off switchs on the systems’ suction units. If…
Editor's Note The Food and Drug Administration’s (FDA’s) Vaccines and Related Biological Products Advisory Committee on June 28 recommended updating the composition of COVID-19 booster vaccines to target the Omicron variant. The Committee noted that booster doses consisting of an Omicron monovalent or bivalent vaccine could improve neutralizing antibody responses…
Editor's Note The Joint Commission, on June 28, announced a new Quick Safety advisory on “Managing packaged sterile supplies and devices.” The advisory provides guidance to keep patients safe from infection and harm from expired or compromised supplies and devices and urges healthcare workers (HCWs) to pay close attention to…
Editor's Note The Food and Drug Administration (FDA) on June 28 identified the recall by GE Healthcare of its CARESCAPE R860 Ventilator as Class I, the most serious. The recall was initiated because the ventilator backup batteries, including replacement backup batteries, may run out before they are expected to do…
Editor's Note The Food and Drug Administration (FDA) on June 23 identified the recall by Baxter Healthcare Corporation of its Volara system with in-line ventilator adaptor as Class I, the most serious. The Volara system is used to clear mucus out of airways, expand lungs, and treat or prevent pulmonary…
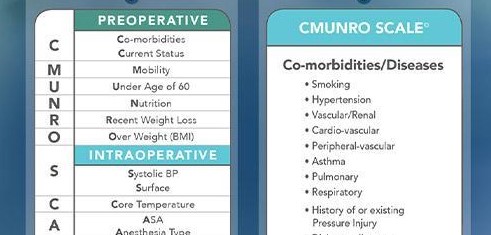
After a journey of more than 20 years, validity and reliability studies for the Munro Pressure Injury Risk Assessment Scale for Perioperative Patients (Munro Scale) show that the Munro Scale works. The Munro Scale, created by Cassendra A. Munro, PhD, MSN, RN, RNFA, CNOR, is the first standard tool that…
Editor's Note The Food and Drug Administration (FDA), on June 23, identified the recall by Medtronic of its HeartWare Ventricular Assist Device (HVAD) batteries as Class I, the most serious. The recall for a single lot (Model Number:1650DE) of HVAD batteries was initiated because of welding defects that allow separation…
Editor's Note The Joint Commission on June 22 announced a new “R3 Report—Issue 36: New Requirements to Reduce Health Care Disparities,” which examines the rationale and references behind new and revised requirements to reduce healthcare disparities in accredited organizations, effective January 1, 2023. Among the new and revised requirements: A…
Editor's Note The Food and Drug Administration (FDA) on June 21 identified the recall by Draeger, Inc, of one lot (LT2103) of its SafeStar 55 Breathing System Filters as Class I, the most serious. The Breathing System Filter is used as part of a ventilator when a patient is under…
Editor's Note This study by researchers from Cornell University finds that nasopharyngeal swabs taken from back inside the nostril were more effective at detecting COVID-10 than saliva tests or swabs just inside the nostril or under the tongue. Comparing different samples from 77 patients, the researchers found that: Nasopharyngeal samples…