
When Phyllis Quinlan, PhD, RN, NPD‑BC, founded MFW Consultants in 1994, she was already on her third career pivot. The former social‑work intern turned emergency‑trauma nurse discovered that the high-stakes of the emergency department (ED), coupled with her human‑behavior insights from sociology and psychology degrees, gave her a rare vantage point on how…

Editor's Note The Accreditation Association for Ambulatory Health Care (AAAHC) released its v44 Standards, introducing new guidance designed to strengthen patient-focused care, streamline compliance, and align with evolving state regulations, an August 18 AAAHC announcement reports. As detailed in the news release, the v44 Standards, effective December 16, 2025, were…

Editor's Note University of California San Francisco (UCSF) became the first university to certify medical students as robotic surgery bedside assistants, giving future surgeons unprecedented hands-on training in a rapidly growing field, an August 25 UCSF news release reports. The program reportedly positions learners alongside attending surgeons, nurses, and scrub…

Editor's Note Artificial disc replacement (ADR) in the cervical spine can be performed safely, efficiently, and cost-effectively in outpatient/ambulatory surgery centers (ASCs), Ortho Spine News July 31 reports. The article details research from Steven J. Girdler, MD, of DISC Surgery Center, who reviewed 6 years of data from 1,043 patients…
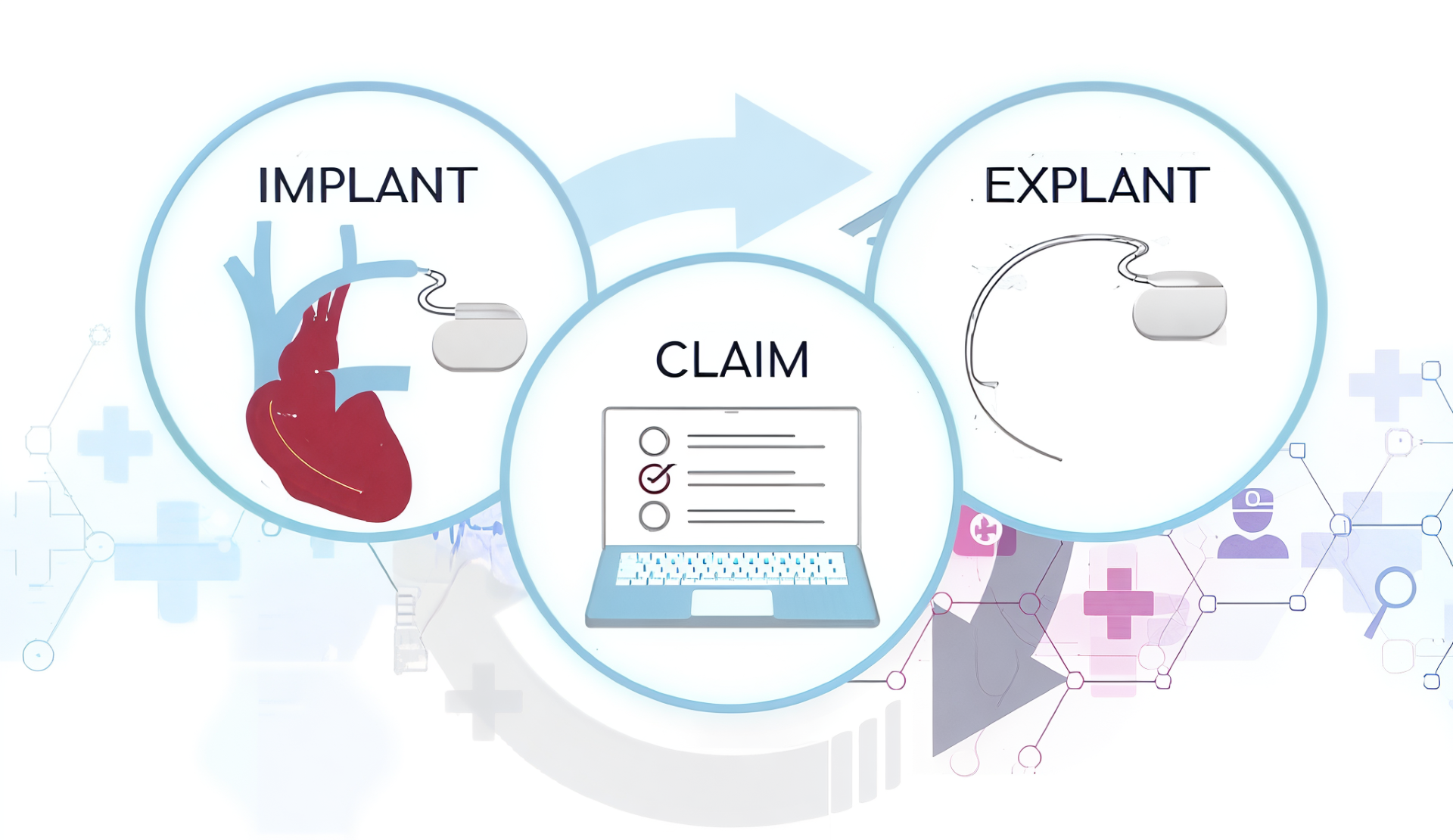
Ambulatory surgery centers (ASCs) are taking on more high-acuity, same-day procedures than ever before. This growth is driven by evolving clinical protocols, cost-conscious reimbursement strategies, and expanded capabilities in outpatient care. Specialties like orthopedics, spine, ophthalmology, and cardiovascular care are moving more complex cases—and the implants that come with them—out…
Once limited to hospital inpatient settings, total joint surgery is increasingly common at ambulatory surgery centers (ASCs) across the US. What is not so common is performing these complex procedures without the benefit of an onsite sterile processing department (SPD). And yet, that is exactly what we have accomplished at…

Editor’s Note: This page is a companion piece to the main article, Centralized sterile processing cuts costs, complexity for four ASCs. Implementing offsite sterilization is a major project. At Total Joint Specialists, our journey began gradually, growing in scope over time as the team became comfortable with the process…

Editor's Note The FDA has issued a Class 1 recall—the most severe category indicating risk of serious injury or death—for Draeger Filter SafeStar 90 Plus (MP05785), Filter SafeStar 55 Plus (MP05790), Filter SafeStar 60A Plus (MP05795), and Filter/HME TwinStar HEPA Plus (MP05801) breathing system filters, affecting all lot numbers. Draeger…

Editor's Note Hospital CEO turnover climbed 3% in the first half of 2025, with organizations shifting toward interim appointments as financial and regulatory pressures intensify leadership changes, according to an August 5 article in HealthLeaders. Citing data from Challenger, Gray & Christmas, the outlet reports that hospitals recorded 68 CEO…

Editor's Note Running effective emergency drills in ambulatory surgery centers (ASCs) requires strategic resource use, role clarity, and consistent adaptation, Texas Ambulatory Surgery Center Society July 8 reports. Drawing on insights from Laura Schneider, senior clinical director at AMSURG, the piece outlines practical steps to ensure ASCs can respond effectively…