 Supply Chain/Technology
Supply Chain/Technology
Proposed federal rule would require hospitals, health systems to report cyberattacks

Editor's Note The Department of Homeland Security's Cybersecurity and Infrastructure Security Agency (CISA) proposed a new rule that would require hospitals and health systems to report any cyberattacks or ransomware attacks to the agency within 72 hours and any ransomware payments within 24 hours. The healthcare sector is one of…
Bioelectric mesh could help track cardiac disease

Editor's Note Although cardiac disease is the leading cause of human morbidity and mortality globally, the complexity of cardiac tissues makes monitoring difficult. To address this issue, a team from University of Massachusetts Amherst and MIT has created a bioelectronic mesh system that can measure movement of cells in cardiac…
Surgical robot specializes in head, neck tumor resection

Editor's Note A team of Johns Hopkins researchers has designed a robot capable of performing the most complex, delicate procedures—including head and neck tumor resection—with accuracy greater than many human surgeons. As reported by the institution on March 18, ASTR: the Autonomous System for Tumor Resection is an autonomous, dual-arm,…
Study: UV-C light effectively disinfects non-sterile, high-touch surfaces

Editor's Note Although many studies have focused on infection transmission within the operating room, authors of research published in the March issue of the Journal of Infection Control focused their study of UV-C light disinfection on non-sterile hubs of patient care—in this case, high-touch surfaces within an academic endoscopy unit.…
Forced-air device outperforms standard endoscope drying practices, study shows

Editor's Note Authors of a recent study evaluating the effectiveness of a forced-air drying system for endoscopes argue that the results reinforce the need to re-evaluate standard drying practices. Findings were published February 24 in the American Journal of Infection Control. Wet environments resulting from inadequate drying practices can result…
Swifty’s singing keeps brain surgery on track

Editor's Note Staying awake during brain surgery to sing Taylor Swift songs helped ensure the best possible outcome for Selena Campione, a 36-year-old teacher from Stanhope, New Jersey who recently had a tumor removed at Jersey Shore University Medical Center. As detailed in a March 21 report from People, neurooncologist…
Surgical scrub evolution and the future of smart medical attire

For surgeons and other medical professionals, what to wear to work is more than just an afterthought. Over the decades, surgical scrubs have undergone a significant transformation, evolving from simple, functional garments to sophisticated attire that prioritizes both comfort and infection control. They are not merely clothing but a vital…
How gamification improves OR training, outcomes
Takeaways From mobile phone apps to immersive virtual reality surgery experiences, digital tools make healthcare training more interactive, more engaging, and more fun for everyone from nurses to surgeons. Modern procedures—including minimally invasive techniques and robotic surgery—require 50 to 100 cases for a surgeon to reach a safe proficiency level…
AI’s pivotal role in transforming OR efficiency, now and in the future
Next-Gen Disruptors: Technologies Transforming the OR Installment #1, presented by LeanTaaS Surgical procedures are necessary steps in patient journeys—no one wants to have surgery, but most people will need it at some point for care continuity and better quality of life. To best support positive outcomes, optimal recovery periods, and…
Procedural sedation analgesia considerations for ASC leaders
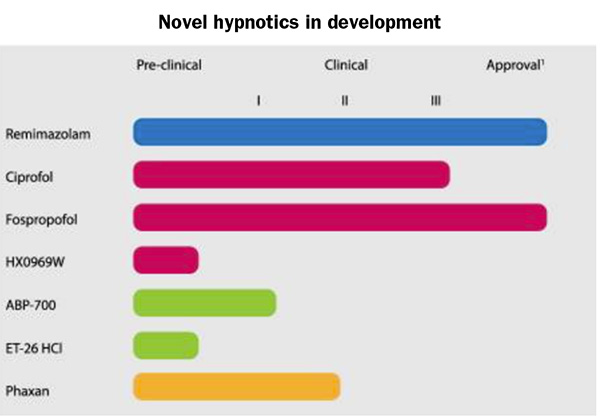
The promise of quicker recovery and fewer complications from sedation, anesthesia, and pain management have drawn clinicians and patients alike to procedures performed in ambulatory surgery centers (ASCs) and other outpatient settings. However, sedation, anesthesia, and analgesia add their own risks to those of the procedure itself. Understanding the latest…

 Free Daily News
Free Daily News